- Blog
- Youtubers life 2 characters likes and dislikes
- Mh4u kushala vise x
- Predator helios 500
- Beach cleanup near me
- I love you quotes
- Racerender alternative
- Synonym for follow up
- Wolf children
- Broken stones paul weller
- Add shareplus 60
- Antimony metal nonmetal or metalloid
- Chronosync slow when locked
- Cyberduck for mac
- Call of duty black ops zombies apk download 4share

A mixing experiment under controlled laboratory conditions tended to support equilibration between the dissolved phase and particles present in the maximum turbidity zone that may induce both As release and removal in the estuarine freshwater reaches, with As distribution evolving toward a distinct partition coefficient ( K d) value for increasing SPM concentrations ( Masson et al., 2009). Arsenic will adsorb more readily to SPM than antimony and, while there was evidence for bioaccumulation of As by freshwater macrophytes, there was no such evidence for Sb. They also found that, while the removal processes affecting Sb will also affect As, the inverse did not necessarily apply, meaning that processes effective in the removal of Sb are also effective at removing As, but those effective at removing As may not necessarily be effective at removing Sb. The most significant potential removal process was identified as adsorption of Sb onto suspended particulate material (SPM). Wilson and Webster-Brown (2009) demonstrated that in such river systems Sb exhibited a mainly conservative behavior, and seasonally variable dilution was found to be the most important control on Sb concentrations. Geothermal systems are a source of Sb into some freshwaters of New Zealand. They might end up as deposits in the marine environment from human use in particular, they are major waste products from the nonferrous metal industry but also from oil and coal production and the phosphate fertilizer industry.Īntimony is an element that is becoming of increasing concern as an environmental contaminant. Humans use these chemicals in treatments for agriculture (fungicides, herbicides, and insecticides), wood protection, and the glass and alloy industry. Such elements are found in the form of sulfide, arsenide, and arseno-sulfide. Metalloids include boron (Bo), silicium (Si), germanium (Ge), arsenic (As), antimony (Sb), and tellurium (Te). Mazik, in Treatise on Estuarine and Coastal Science, 2011 8.04.3.1.2 Other mineral micropollutants
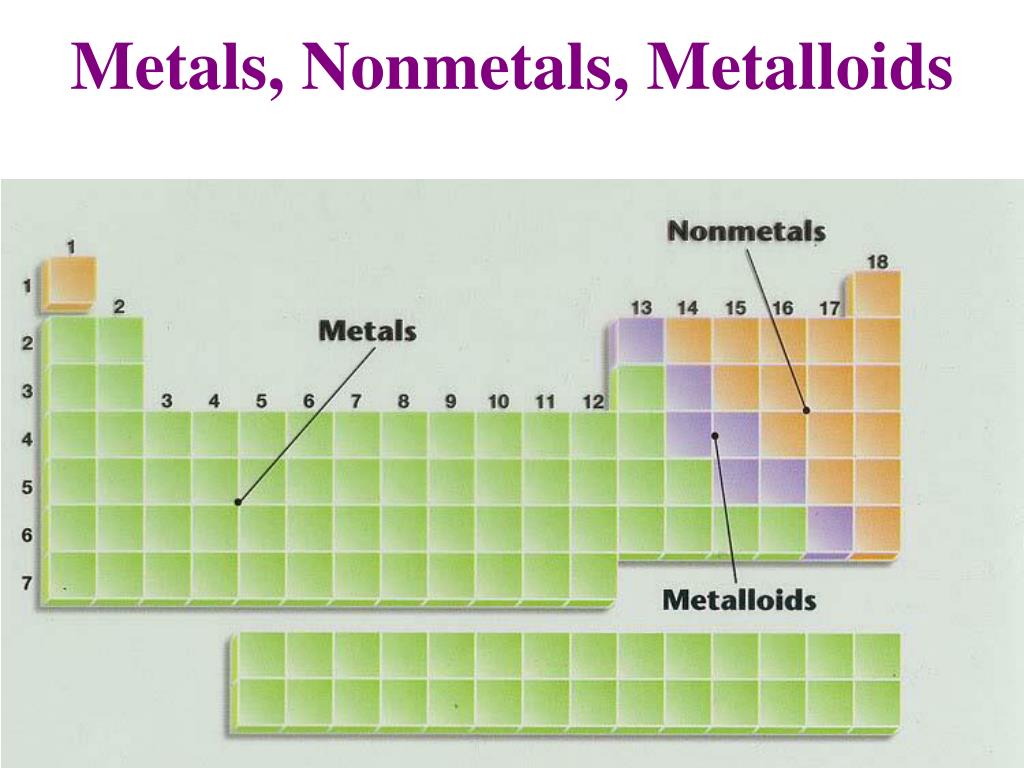
What are Metallaoids list the properties metalloids? Is antimony Sb a metal nonmetal or metalloid?Īntimony (Sb), a metallic element belonging to the nitrogen group (Group 15 of the periodic table). Germanium (Ge), a chemical element between silicon and tin in Group 14 (IVa) of the periodic table, a silvery-gray metalloid, intermediate in properties between the metals and the nonmetals. Silicon and Germanium have properties of both hence they are classified as metalloids. Why are silicon and germanium classified as metalloids?Ī metalloid is any chemical element which has properties in between those of metals and nonmetals, or that has a mixture of them. The ability of the metalloids to conduct electricity and heat is far better than the nonmetals, for example, diamond, which are insulators. Why metalloids are used as semiconductors?Ī number of the metalloids are semiconductors as they have intermediate conductivity which is temperature dependent. On the periodic table, the elements colored yellow, which generally border the stair-step line, are considered to be metalloids. Metalloids can also be called semimetals. Why some elements are called metalloids?Ī metalloid is an element that has properties that are intermediate between those of metals and nonmetals. Antimony is used in the electronics industry to make some semiconductor devices, such as infrared detectors and diodes. In its metallic form it is silvery, hard and brittle. Is antimony a metalloid or metal?Īntimony is a semi-metal. These elements look metallic however, they do not conduct electricity as well as metals so they are semiconductors. The metalloids are boron, silicon, germanium, arsenic, antimony, and tellurium.
#Antimony metal nonmetal or metalloid series#
8 What are Metallaoids list the properties metalloids?Ī series of six elements called the metalloids separate the metals from the nonmetals in the periodic table.7 Is antimony Sb a metal nonmetal or metalloid?.5 Why are silicon and germanium classified as metalloids?.4 Why metalloids are used as semiconductors?.3 Why some elements are called metalloids?.
- Blog
- Youtubers life 2 characters likes and dislikes
- Mh4u kushala vise x
- Predator helios 500
- Beach cleanup near me
- I love you quotes
- Racerender alternative
- Synonym for follow up
- Wolf children
- Broken stones paul weller
- Add shareplus 60
- Antimony metal nonmetal or metalloid
- Chronosync slow when locked
- Cyberduck for mac
- Call of duty black ops zombies apk download 4share
